Gasses
- Avogadro and Gasses
- Combined Gas Law
- Dalton's Law of Partial Pressure
- Gasses
- Ideal Gas Law
- Measuring a Gas
- Real Gasses
Postulates of Gas Behaviour
List
- A gas is composed of a very large number of molecules
- These molecules are separated by a large distance relative to their own size
- A gas is composed of a large number of molecules in ceaseless motion
Maxwell-Boltzmann Speed Distribution
Definition
The speeds of ideal gas molecules obey the Maxwell-Boltzmann speed distribution:
also written as:
where:
Average Speeds
Most Probable Speed
Formula
Find the maximum value of the distribution function by setting the derivative to 0:
Average Speed
Formula
Find the expected value of the distribution function by evaluating the integral:
Root-Mean-Square Speed
Formula
Evaluate the integral
Effects of Temperature and Mass
Info
- Tempetrature
- Higher temperature results in a broader range of speeds
- Distribution shifts towards higher speeds as temperature increases
- Mass
- Lower molecular weight results in a broader range of speeds
- Distribution shifts towards higher speeds for molecules with a lower molecular weight
Figure
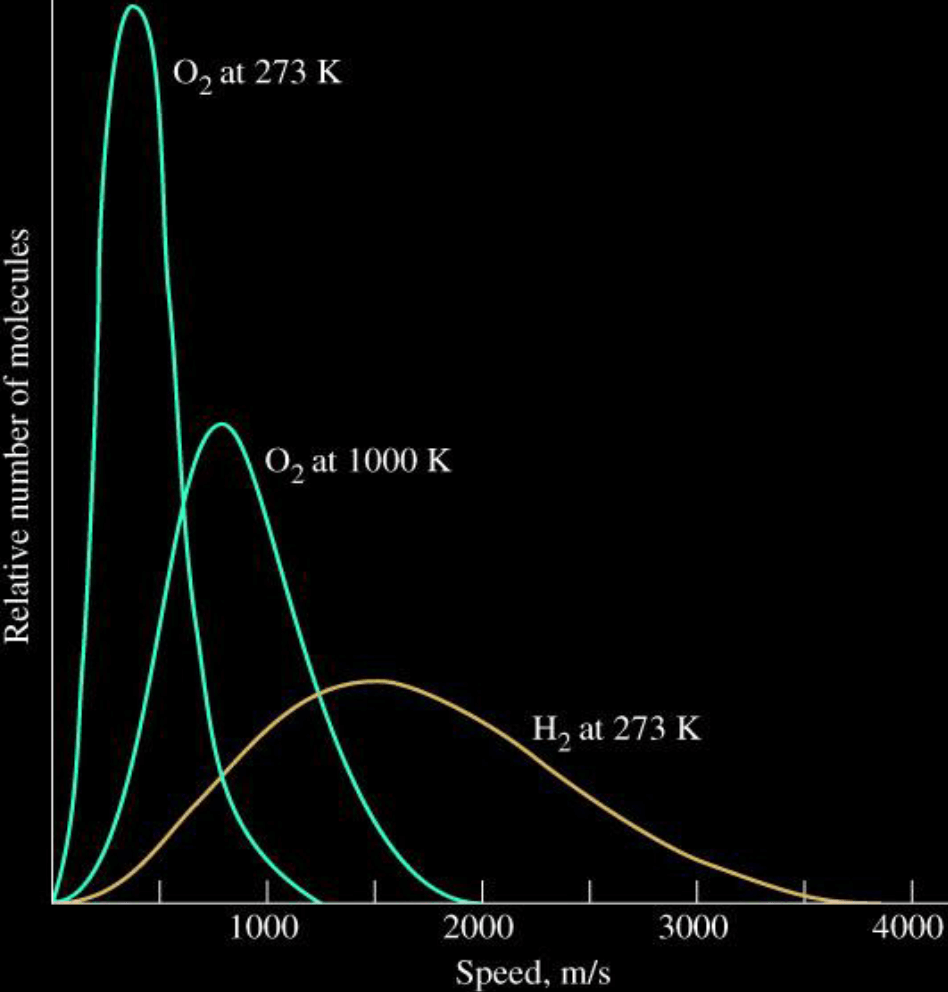
Kinetic-Molecular Theory
List
Postulates:
- A gas is composed of a very large number of molecules in ceaseless random straight-line motion
- These molecules are separated by a large distance relative to their own size
- The molecules behave like hard spheres that undergo perfectly elastic collisions with one another and the walls of the container
- There are no forces of attraction or repulsion between the molecules
Example
Two containers of argon gas are illustrated below. Both containers contain the same number of moles of argon and are at the same temperature.
- Is the root-mean-square speed of the argon the same in both containers?
Yes - Is the frequency with which the argon atoms collide with the walls the same in both containers?
No - Is the average force of each collision between an argon atom and the wall the same in both containers?
Yes
Gas Density
Formula
where: